
Assessment of Disease Profiling
Disease-specific human iPSC-derived cardiomyocytes (hiPSC-CMs) enable physiologically relevant modeling of genetically inherited heart diseases, improving our understanding of impaired electrical and contractile function.
Unlike animal models, patient-derived hiPSC-CMs offer higher translational relevance and are well suited for disease characterization and drug screening. The xCELLigence RTCA Cardio and xCELLigence RTCA CardioECR systems measure integrated cardiomyocyte function, enabling robust assessment of cardiac disease phenotypes and evaluation of potential therapeutics.
Application Note:
Assessment of hiPSC Cardiomyocyte Disease Models
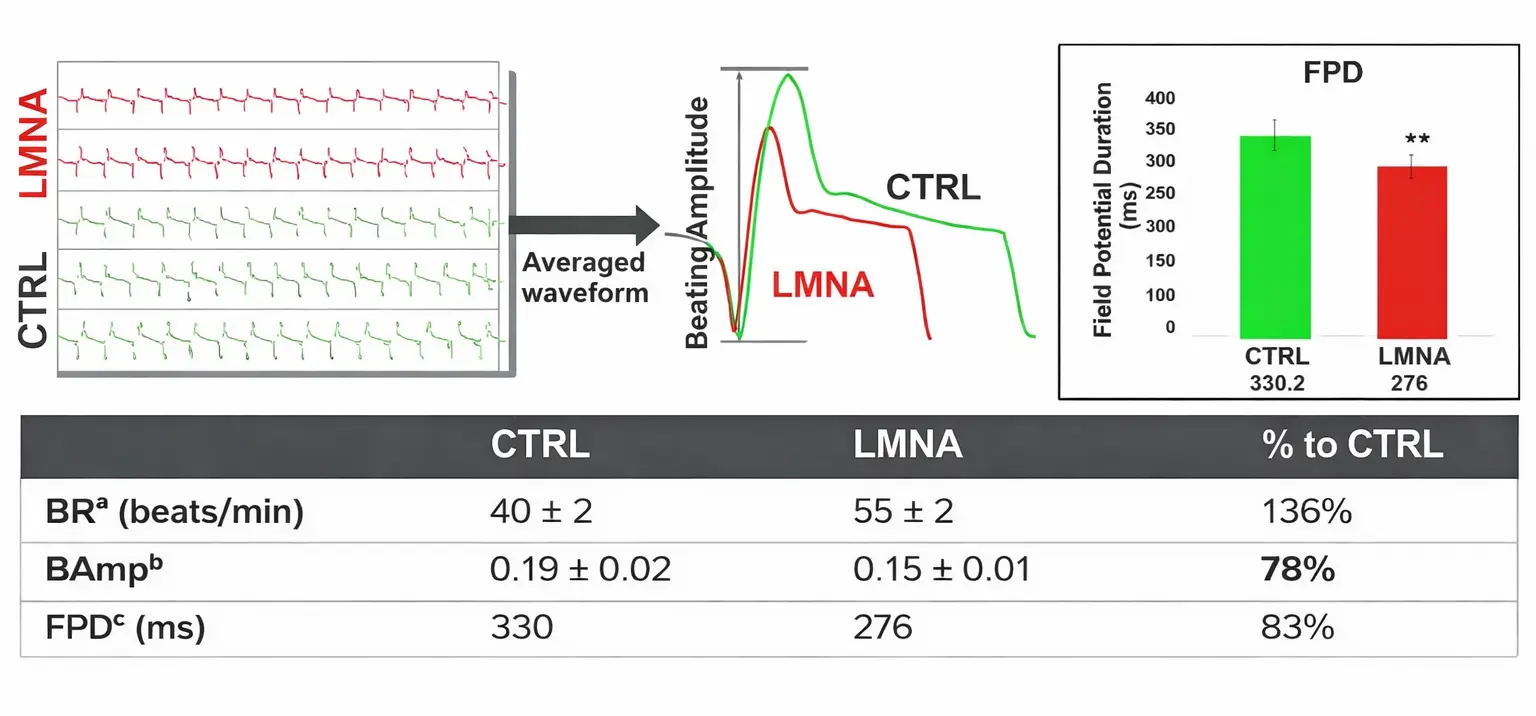
Functional Profile of LMNA Cardiomyopathy and Isogenic Control Cardiomyocytes
Contractile and electrical activities of LMNA and control (CTRL) cardiomyocytes were assessed 12 days post-seeding, when consistent and robust contractile and electrical signals were observed. Data are presented as mean ± SD (N = 24).
Abbreviations:
BRa = beating rate; Ampb = beating amplitude; FPDc = field potential duration measured during electrical pacing at 1 Hz.
Statistical significance: **P < 0.01.
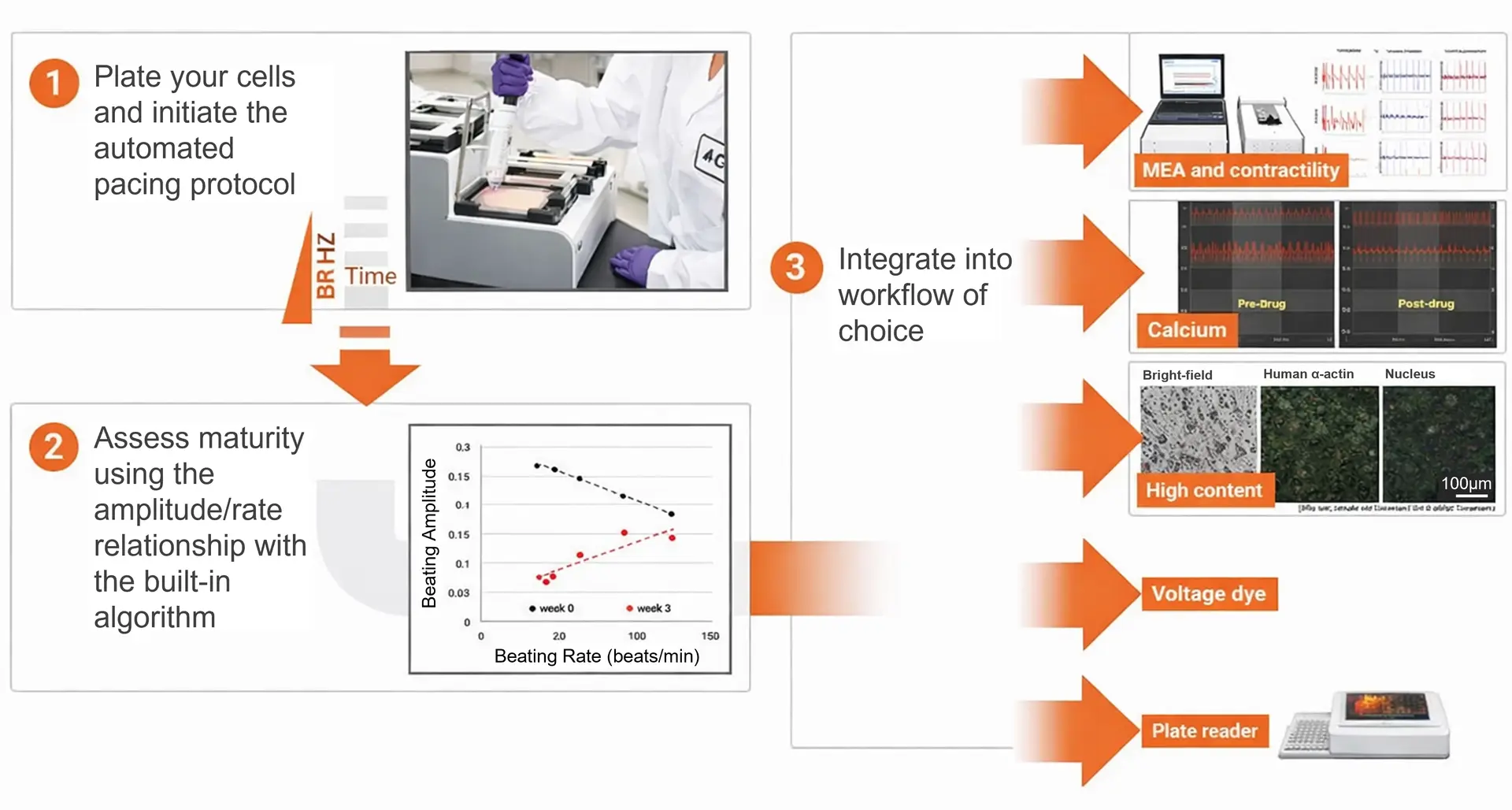
Overcome research hurdles and uncover disease mechanisms
Simplify your workflow with label-free analysis. Just seed your cells, monitor in real time, and evaluate disease mechanisms as well as drug efficacy and toxicity in cardiomyocytes.
Application Note:
Cardiomyocyte Maturation Using the xCELLigence RTCA ePacer
Literature
Application Notes
Assessment of Cardiomyocyte Disease Models
A Real-Time Impedance Analysis Instrument for Cardiac Safety and Toxicity Assessment
The xCELLigence RTCA CardioECR System
Functional Maturation of
Human-Induced Pluripotent Stem
Cell-Derived Cardiomyocytes
Publications
- Precision Health Resource of Control iPSC Lines for Versatile Multilineage Differentiation. Hildebrandt MR, Reuter MS, Wei W, Tayebi N, Liu J, Sharmin S, Mulder J, Lesperance LS, Brauer PM, Mok RSF, Kinnear C, Piekna A, Romm A, Howe J, Pasceri P, Meng G, Rozycki M, Rodrigues DC, Martinez EC, Szego MJ, Zúñiga-Pflücker JC, Anderson MK, Prescott SA, Rosenblum ND, Kamath BM, Mital S, Scherer SW, Ellis J. Stem Cell Reports. 2019 Dec 10;13(6):1126-1141. (The Hospital for Sick Children, Canada)
- Parabolic, Flight-Induced, Acute Hypergravity and Microgravity Effects on the Beating Rate of Human Cardiomyocytes. Acharya A, Brungs S, Lichterfeld Y, Hescheler J, Hemmersbach R, Boeuf H, Sachinidis A. Cells. 2019 Apr 14;8(4). pii: E352. (University of Cologne, Germany)
- Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes as Models for Cardiac Channelopathies: A Primer for Non-Electrophysiologists. Priyanka Garg, Vivek Garg, Rajani Shrestha, Michael C. Sanguinetti, Timothy J. Kamp and Joseph C. Wu. Circ Res. 2018 Jul 6;123(2):224-243. (Stanford Cardiovascular Institute, USA)
- MLP and CARP are linked to chronic PKCα signalling in dilated cardiomyopathy. Lange S, Gehmlich K, Lun AS, Blondelle J, Hooper C, Dalton ND, Alvarez EA, Zhang X, Bang ML, Abassi YA, Dos Remedios CG, Peterson KL, Chen J, Ehler E. Nat Commun. 2016 Jun 29;7:12120. (University of California, San Diego. USA)
- Integrin-linked kinase mediates force transduction in cardiomyocytes by modulating SERCA2a/PLN function. Traister A, Li M, Aafaqi S, Lu M, Arab S, Radisic M, Gross G, Guido F, Sherret J, Verma S, Slorach C, Mertens L, Hui W, Roy A, Delgado-Olguín P, Hannigan G, Maynes JT, Coles JG. Nat Commun. 2014 Sep 11;5:4533. (Hospital for Sick Children, Canada)
- Study familial hypertrophic cardiomyopathy using patient-specific induced pluripotent stem cells. Han L, Li Y, Tchao J, Kaplan AD, Lin B, Li Y, Mich-Basso J, Lis A, Hassan N, London B, Bett GC, Tobita K, Rasmusson RL, Yang L. Cardiovasc Res. 2014 Nov 1;104(2):258-69. (University of Pittsburgh School of Medicine, USA)A Real-Time Impedance Analysis Instrument for Cardiac Safety and Toxicity Assessment
For Research Use Only. Not for use in diagnostic procedures.

