Cardio Safety Toxicology
Cardiac liability is a leading cause of late-stage drug development failures, with cardiotoxicity accounting for roughly one-third of drug withdrawals between 1990–2006. Traditional preclinical models often lack predictive power, creating a need for physiologically relevant screening systems early in drug discovery.
Human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) provide a robust platform to assess drug-induced cardiotoxicity and pro-arrhythmic risk.
The xCELLigence RTCA CardioECR system enables multiplex detection of contractile activity, electrical function, and cell viability in a single, sensitive assay - helping researchers to identify cardiotoxic risks earlier and more accurately.
Application Note:
Real-time analysis for cardiac safety and toxocity assessment
Assessment of Contractile Related Toxicities
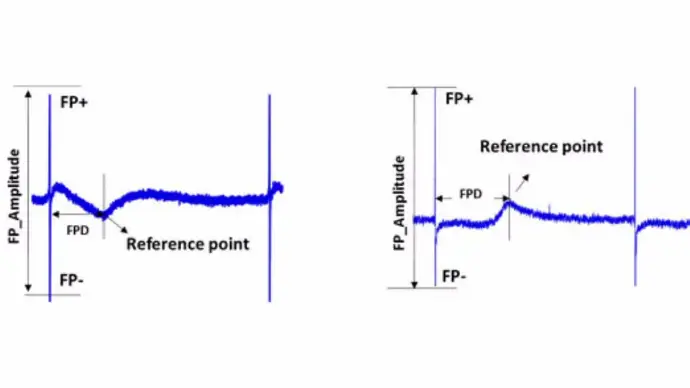
The existing methods for assessment of contractile activities, such as Langendorff heart assays and other ex vivo whole organ assays are labor intensive, low throughput, and expensive. Alternatively, animal studies can also be conducted, but the predictivity of these assays and translational potential to humans is always an important consideration in addition to the ethical considerations of using animals. Functional monitoring of cardiomyocyte contractility using impedance readout (see figure to left) provides incisive information about compound effects on the contractile machinery.

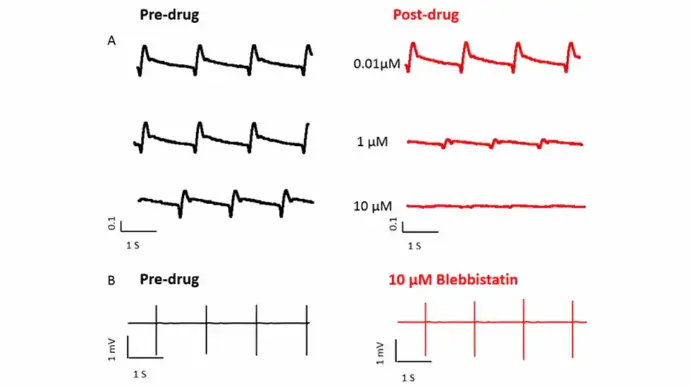
Case study: Blebbistatin, a myosin heavy chain inhibitor, decouples excitation and contraction coupling
Blebbistatin reduced beating amplitude in a dose-dependent manner but did not alter the field potential signal even after exposure to 10 µM blebbistatin.
(A) 5 seconds of beating amplitude traces obtained from iCell cardiomyocytes before (black traces) and after (red traces) blebbistatin treatment at 0.01, 1, and 10 µM. The compound addition was performed in one dose one well fashion.
(B) 5 seconds of FP traces obtained before (black trace) and after 10 µM of blebbistatin treatment.
Assessment of Acute & Long-term Ion Channel Toxicities
Assays that more directly assess possible pro-arrhythmic effects of compounds utilize whole animal hearts or Purkinje fibers and are designed to assess action potential duration. While these assays are considered to be more predictive of arrhythmia, they have higher negative predictivity rate and can be low throughput and technically demanding. Human iPSC-CMs provide a physiologically relevant model that can be used to assess potential toxicity of pharmaceutical compounds or integrated ion channel activity.
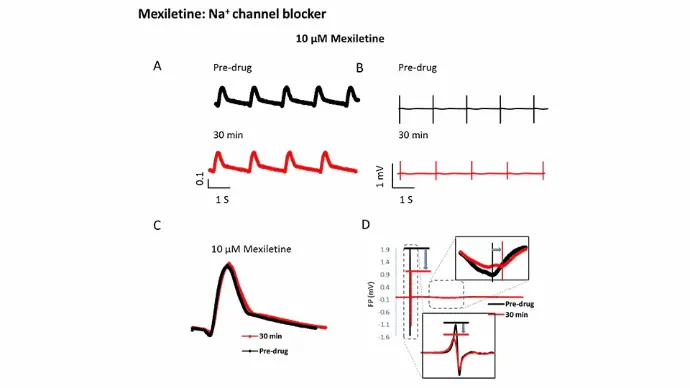
Na+ channel
Mexiletine, a Na+ channel blocker, induced reduction of field potential (FP) spike, suggesting the blockade of sodium channel.
5 seconds of impedance (IMP) (A) and field potential (FP) (B) signals of iCell CMs before (black traces) and after (red traces) exposure to 10 µM Mexiletine. The raw IMP and FP traces were then further calculated to single averaged waveforms (C and D).
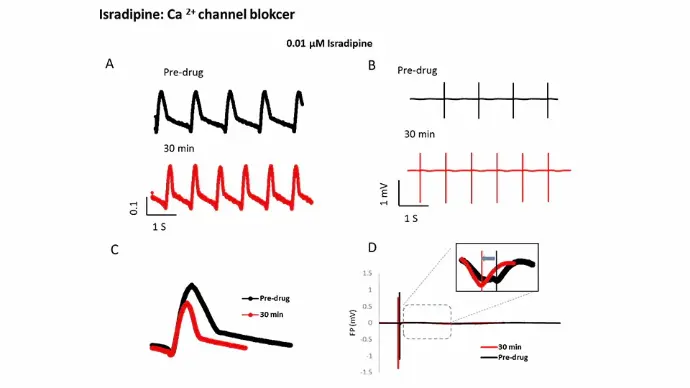
Ca2+ channels
Isradipine, a Ca2+ channel blocker, induced field potential duration (FPD) shortening and decrease in IMP amplitude/beating amplitude (BAmp).
5 seconds of impedance (IMP) (A) and field potential (FP) (B) signals of iCell cardiomyocytes before (black traces) and after (red traces) exposure to 0.01 µM isradipine. The raw IMP and FP traces were then further calculated to single averaged waveforms (C and D).
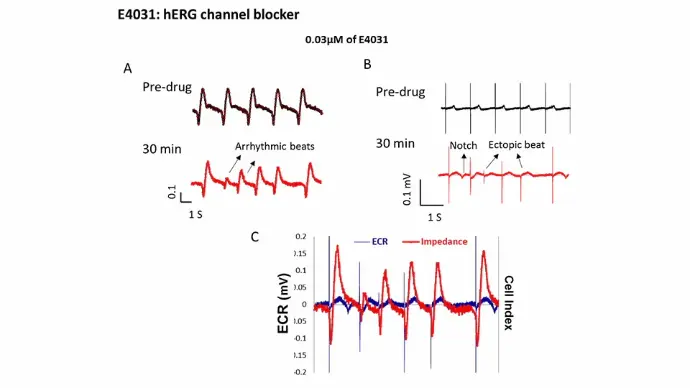
hERG channel
E4031, a hERG channel blocker, induced EAD-like FP signal and arrhythmic beating on beating amplitude traces.
5 seconds of impedance (beating amplitude) (A) and field potential (FP) (B) signals of iCell cardiomyocytes before (black traces) and after (red traces) exposure to 0.03 µM E4031. The overlay of IMP and FP traces (C) 30 min post-drug.
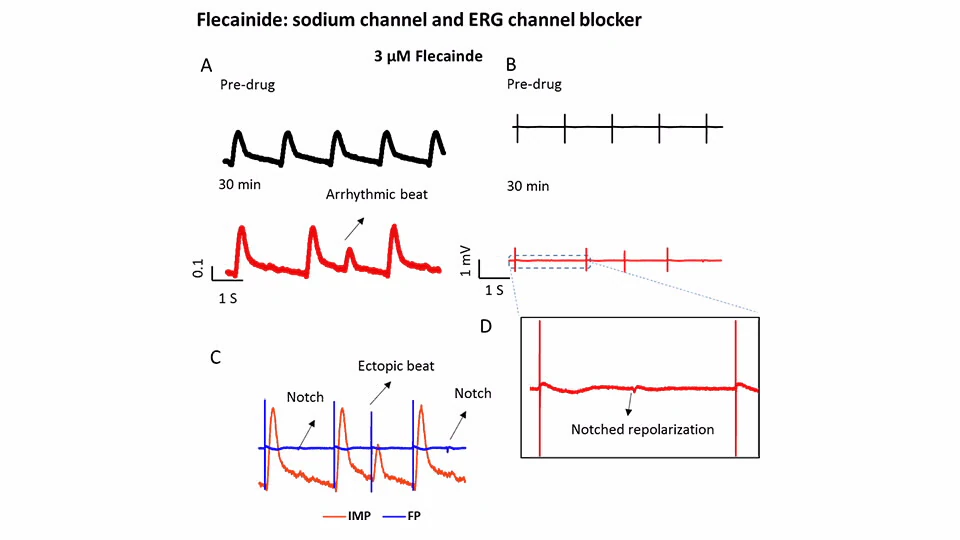
Multi-ion channel
Flecainide: a multichannel blocker, which inhibits Na+ and hERG channels. Flecainide induced FP spike reduction and EAD-like (notch and ectopic beat) on FP traces and arrhythmic beating on IMP traces.
5 seconds of impedance (IMP) (A) and field potential (FP) (B) signals of iCell cardiomyocytes before (black traces) and after (red traces) exposure to 3 µM flecainide. (C) The overlay of IMP and FP traces (C) 30 min post-drug. (D) Magnified in FP trace
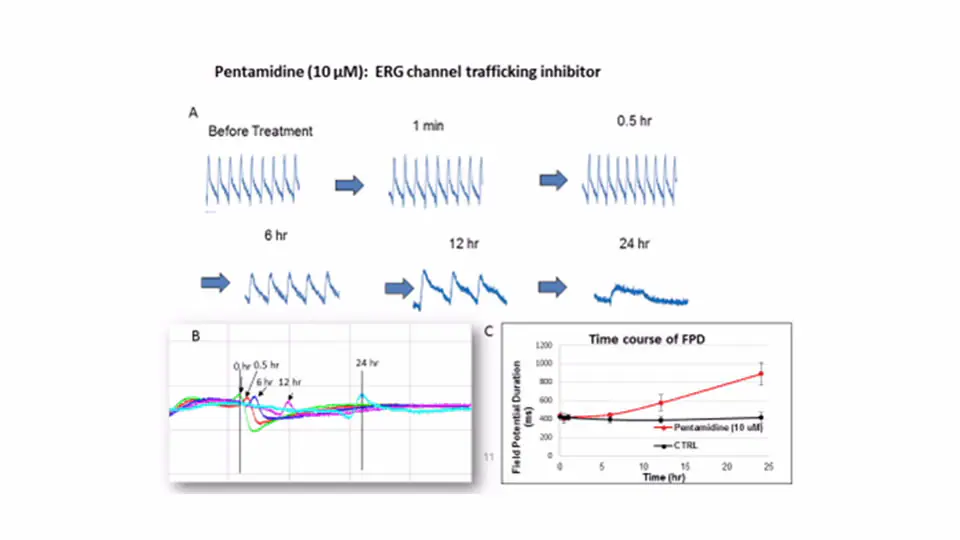
Pentamidine, a hERG channel trafficking inhibitor, showed delayed FPD and contractility period duration
(A)15 seconds of IMP traces obtained from iCell cardiomyocytes before and after pentamidine treatment at the indicated time points.
(B) The expanded FP traces recorded before and after 10 µM pentamidine treatment at the indicated time points.
(C) Time course of FPD measurement over the course of 10 µM pentamidine treatment.
Assessing Chemotherapy-Induced Cardiotoxicity (Cardio Oncology)
Chemotherapeutic drugs, including anthracyclines and certain kinase inhibitors, can damage cardiomyocyte structure. Traditional end-point assays may miss transient effects, making early detection challenging.
The xCELLigence CardioECR system uniquely measures structural integrity, viability, ion channel activity, and contractility simultaneously - offering a comprehensive, sensitive platform for early cardiotoxicity screening.
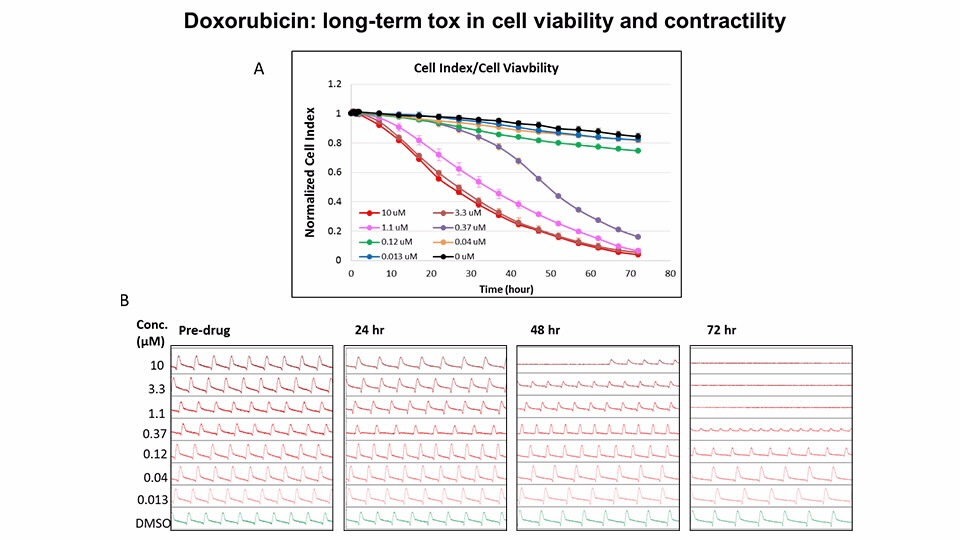
Assessment of Anthracycline Toxicity
Doxorubicin, an anthracycline and chemotherapy drug, is known to cause cardiac toxicity
(A) Overall cell index change over the course of 72-hour treatment.
(B) 15 seconds of impedance traces obtained from iCell cardiomyocytes before and after doxorubicin treatment at the indicated time points.
Doxorubicin induced overall cell index decrease in a dose and time-dependent manner, suggesting a loss of cell viability and/or structural damage to the cells.
Doxorubicin also affected the contractility of cardiomyocytes in a dose and time-dependent manner, which may be related to the loss of viability.
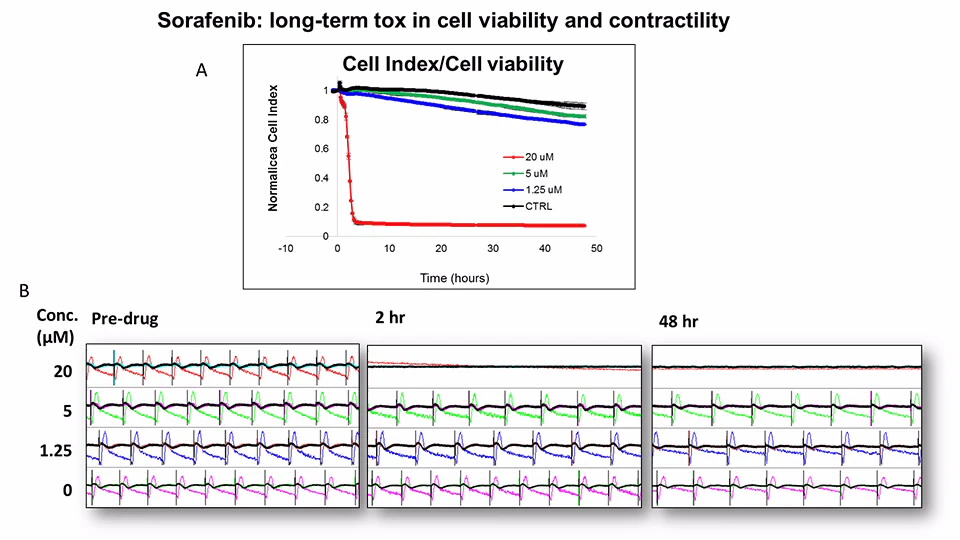
Assessment of Kinase Inhibitor Toxicity
Sorafenib is a well-studied cardiotoxic compound that’s known to cause myocardial infarction and ischaemia.
Sorafenib induced overall cell index decrease in a dose and time-dependent manner. It caused cell quiescence at 20 µM
(A) Overall cell index change over the course of 48-hour treatment.
(B) 15 seconds of impedance traces obtained from iCell cardiomyocytes before and after sorafenib treatment at the indicated time points.
Related Application Notes
A Real-Time Impedance Analysis Instrument for Cardiac Safety and Toxicity Assessment
The Agilent xCELLigence RTCA CardioECR System
Assessment of Cardiomyocyte Disease Models Using the Agilent xCELLigence CardioECR
Functional Maturation of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes
For Research Use Only. Not for use in diagnostic procedures.